Medical & Integrative Medicine
FDA-Cleared, Non-Thermal Therapeutic Platforms
As medical practices continue expanding into procedure-light and adjunctive therapies, there is growing interest in technologies that integrate into established clinical workflows without increasing procedural risk or recovery burden.
Erchonia’s non-thermal laser systems deliver wavelength-specific light energy designed for defined FDA-cleared indications, including musculoskeletal pain, low back pain, dermatological applications, and fat reduction. These platforms are engineered for structured implementation within medical, multidisciplinary, and integrative care environments.
Rather than replacing existing treatment models, Erchonia systems function as adjunctive therapeutic tools — supporting clinical protocols while maintaining operational efficiency.
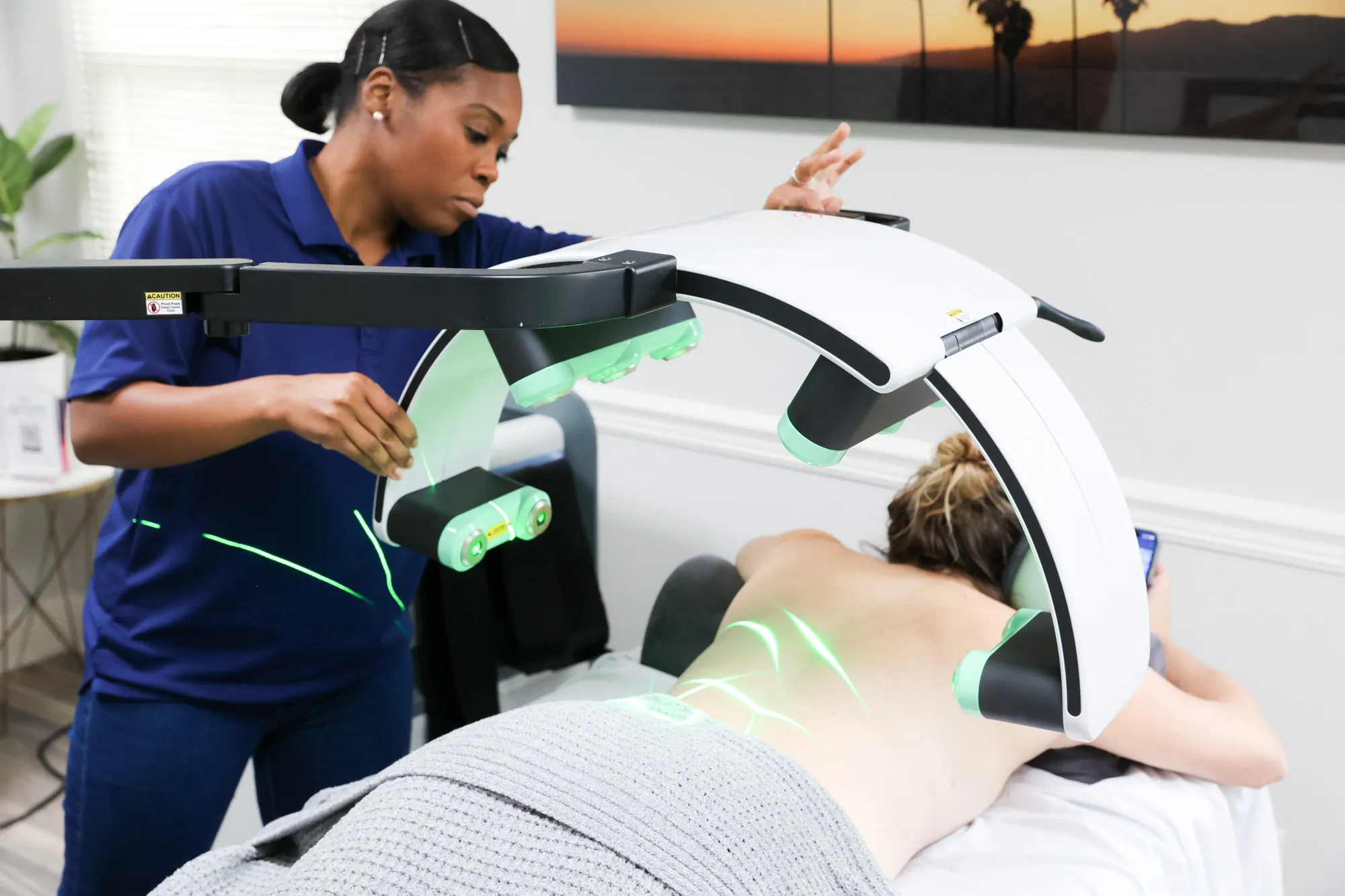
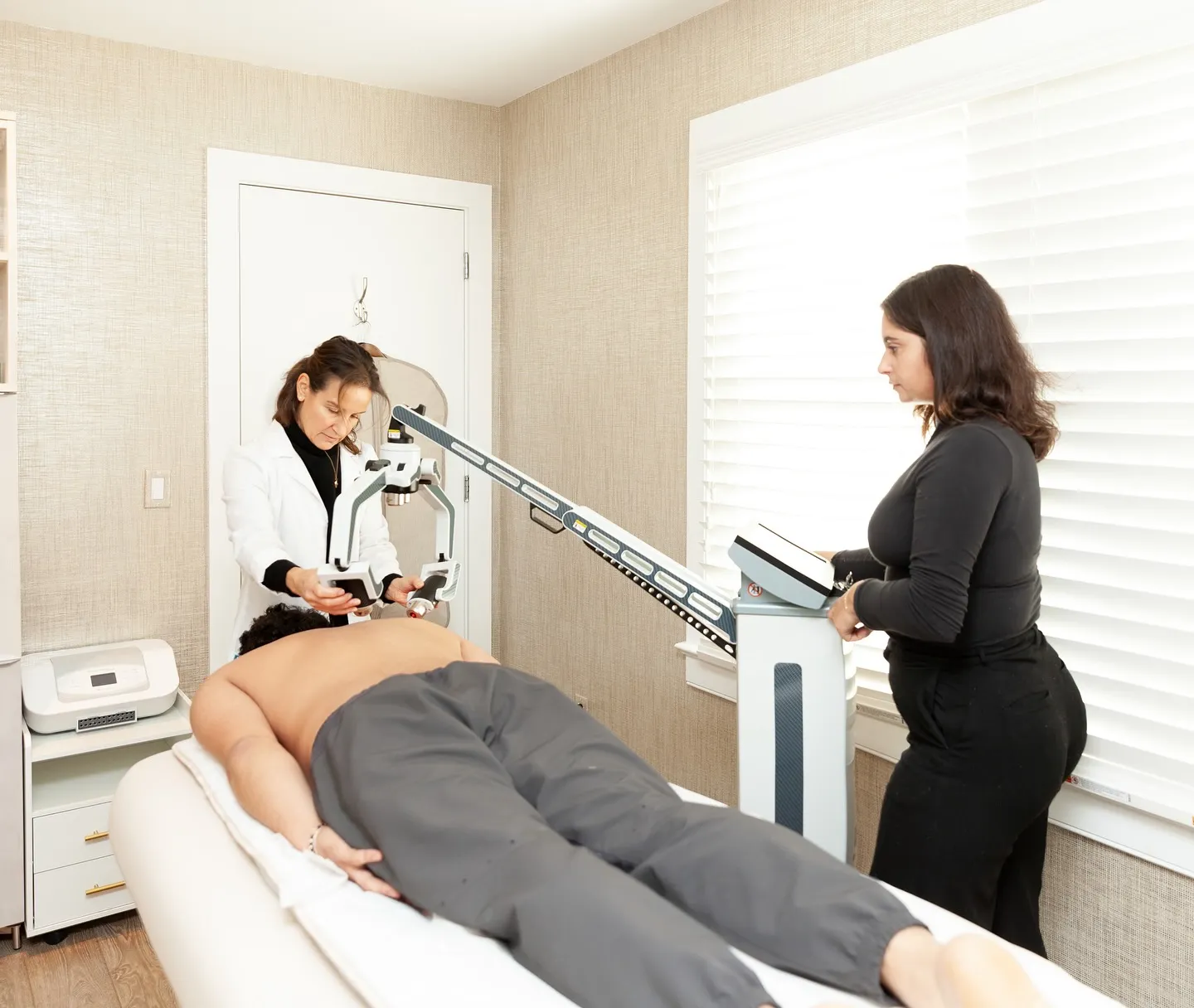
Where Laser Therapy Fits in Medical Practice
Erchonia laser platforms are commonly integrated within:
• Pain management clinics
• Orthopedic and sports medicine practices
• Physical medicine and rehabilitation settings
• Integrative and functional medicine clinics
• Medical weight-loss programs
• Aesthetic-medical hybrid practices
Implementation typically occurs as an adjunct within existing care plans, supporting patient management across defined indications.
Regulatory Alignment & Indication-Based Use
Erchonia became the first company to receive FDA clearance for low-level laser therapy and continues to hold more FDA clearances than all other manufacturers combined within this category.
Platforms within the Erchonia portfolio hold FDA clearances for defined indications including:
• Adjunctive use in providing temporary relief of minor chronic neck and shoulder pain of musculoskeletal origin
• Adjunctive treatment of low back pain (FX Series)
• Adjunctive treatment of overall musculoskeletal nociceptive pain
• Adjunctive treatment of postoperative pain
• Treatment of moderate inflammatory Acne Vulgaris (selected platforms)
• Treatment of Onychomycosis (nail fungus)
• Circumference reduction in individuals with BMI up to 40
• Temporary reduction in the appearance of cellulite
Prescription Use (21 CFR 801 Subpart D). Intended for medical professionals.
